All published articles of this journal are available on ScienceDirect.
Lens-protective Effects of Hesperidin and Naringenin in a Rat Model of Streptozotocin-Induced Diabetes Mellitus
Abstract
Introduction/Objective
Diabetic cataract is driven by hyperglycemia-associated oxidative/nitrosative stress, inflammation, apoptosis, activation of the polyol pathway, and lens protein glycation. Hesperidin (HSN) and Naringenin (NGN) are citrus flavonoids with antioxidant, anti-inflammatory, anti-apoptotic, and antidiabetic activities. This study evaluated their lens-protective effects in streptozotocin (STZ)-induced diabetic rats.
Methods
Male Sprague-Dawley rats with STZ-induced diabetes received hesperidin (100 mg/kg/day, orally) or naringenin (50 mg/kg/day, orally) for 10 weeks. Fasting blood sugar, serum insulin, lenticular malondialdehyde, nitric oxide, total antioxidant capacity, TNF-alpha, IL-6, NF-kB p65, Bax/Bcl-2 ratio, cleaved caspase-3, aldose reductase activity, sorbitol concentration, glycated proteins, and total/soluble proteins were assessed.
Results
STZ significantly increased fasting blood sugar and reduced serum insulin. Diabetic lenses also showed increased levels of malondialdehyde, nitric oxide, TNF-alpha, IL-6, NF-kB p65, Bax/Bcl-2 ratio, cleaved caspase-3, aldose reductase activity, sorbitol, and glycated proteins, with reduced total antioxidant capacity and total/soluble protein levels. HSN and NGN significantly ameliorated these changes versus untreated diabetic rats (p < 0.05).
Discussion
The findings indicate that HSN and NGN mitigate biochemical pathways implicated in diabetic lens injury, including oxidative/nitrosative stress, inflammation, apoptosis, polyol-pathway activation, and protein glycation.
Conclusion
In this STZ-induced rat model, hesperidin and naringenin preserved lens biochemical homeostasis and attenuated diabetes-associated lens injury, supporting their potential as adjunctive candidates for preventing or delaying diabetic lens damage.
1. INTRODUCTION
Diabetes Mellitus (DM) is a global chronic metabolic disorder. According to the latest IDF Diabetes Atlas (11th edition, 2025), approximately 589 million adults worldwide (11.1%) have diabetes [1]. Improper glycemic control results in secondary diabetic complications as nephropathy, retinopathy, neuropathy, lens opacification, and cardiovascular problems. Cataract is one of the earliest secondary diabetic complications. Several clinical and epidemiological studies revealed that the incidence of cataract is three to four-fold higher, and occurs at an earlier age in diabetics than in nondiabetic individuals [2]. Since lens opacification is one of the leading causes of blindness, diabetic cataract represents a major concern in visual impairment management [3]. Therefore, the research for therapeutic strategies to retard and stop the development of cataract in patients with DM is still a challenge.
The precise mechanisms behind the formation of diabetic cataracts remain incompletely understood. Nonetheless, substantial research indicates that several factors contribute significantly to their onset and progression. These include oxidative stress, excessive production of reactive oxygen and nitrogen species (ROS and RNS), weakened antioxidant defenses, lipid peroxidation, inflammation, and apoptosis of lens cells [4, 5]. Moreover, osmotic imbalance and non-enzymatic glycation of proteins contribute to the degeneration of lens fibers and clumping of lens proteins, both of which are hallmarks of diabetic cataract formation [6]. Oxidative stress is also known to upregulate inflammatory mediators such as Tumor Necrosis Factor-Alpha (TNF-α) and Interleukin-6 (IL-6) [7]. Additionally, it activates the Nuclear Factor-Kappa B (NF-κB) signaling pathway, further amplifying inflammatory and apoptotic processes. The downstream activation of apoptotic enzymes, such as caspases, ultimately leads to lens cell death [8].
Hesperidin (HSN) and Naringenin (NGN) are flavonoids predominantly found in citrus fruits and are known for their biological activity. According to existing literature, both compounds have demonstrated the ability to reduce oxidative stress, inflammation, and apoptosis [9, 10]. Moreover, earlier studies have shown that HSN and NGN contribute to improved glycemic control and provide protective effects against diabetes-related complications, such as nephropathy, neuropathy, and cardiovascular disease [11, 12]. Given these properties, HSN and NGN may help prevent lens opacification associated with diabetes. However, to date, their specific lens-protective potential has not been comprehensively examined. Thus, the present study aims to evaluate the protective roles of HSN and NGN in a Streptozotocin (STZ)-induced diabetic cataract model in rats and to investigate the mechanisms underlying their lens-protective effects.
2. METHODS
2.1. Ethical Approval
The research protocol was approved by the Scientific Research Committee, Faculty of Medicine, Al-Baha University (approval number: REC/SUR/BU-FM/2024/21). The study was conducted in accordance with the institutional and national guidelines for the care and use of laboratory animals and is reported in accordance with the ARRIVE guidelines and the 3Rs principles.
2.2. Laboratory Animals
Male Sprague-Dawley rats, approximately 8–10 weeks old and weighing 200 ± 10 g at the start of the experiment, were used in this study. The animals were obtained from the university animal facility. Rats were housed under standard laboratory conditions (24 ± 1°C, 45% relative humidity, 12-h light/12-h dark cycle) with free access to standard chow and tap water ad libitum. Animals were acclimatized for 7 days before the start of the experimental procedures.
2.3. Health Monitoring and Humane Endpoints
Animals were monitored daily throughout the study for general activity, grooming behavior, food and water intake, body-weight changes, and signs of distress or illness. Humane endpoints were predefined as severe lethargy, inability to access food or water, marked and persistent distress, or body-weight loss exceeding the institutional threshold. Animals meeting humane-endpoint criteria were to be removed from the study and humanely euthanized. No animals met the predefined humane-endpoint criteria during the study. At study completion, animals were euthanized by intraperitoneal thiopental (70 mg/kg) after overnight fasting, in accordance with institutional ethical guidance.
2.4. Drugs and Chemicals
HSN, NGN, and Streptozotocin (STZ) powders were purchased from Sigma-Aldrich, USA. HSN and NGN were prepared in a 0.5% carboxymethylcellulose (CMC) solution, and STZ was dissolved in cold citrate buffer (0.1 M, pH 4.5). The doses and routes of administration of HSN, NGN, and STZ used in this work were selected based on previous investigations [11-13].
2.5. DM Induction
A single i.p. injection of STZ at a dose of 45 mg/kg was given to the rats fasted overnight. After 72 h, random blood glucose levels were checked using a glucometer. Rats with values greater than 250 mg/dl were considered diabetic and included in the study.
2.6. Experimental Design
Group sizes (n = 10 rats/group) were chosen based on prior studies using the same STZ-induced diabetic model and similar biochemical endpoints, where 8–10 animals per group were sufficient to detect biologically meaningful differences between diabetic and treated animals.
Rats were randomly divided into 4 equal groups (n = 10, each).
-Group 1 (control) received the vehicle for streptozotocin (0.1 M citrate buffer, pH 4.5) and remained non-diabetic, while Groups 2–4 received streptozotocin to induce diabetes.
-Group 2 rendered diabetics by STZ, and received daily CMC, p.o., for 10 weeks following successful induction of DM.
-Group 3 rendered diabetics by STZ, and received HSN (100 mg/kg/day, p.o.) for 10 weeks following successful induction of DM.
-Group 4 rendered diabetics by STZ, and received NGN (50 mg/kg/day, p.o.) for 10 weeks following successful induction of DM.
After confirmation of diabetes in the STZ-injected rats, diabetic animals were randomly allocated to the diabetic control, HSN-treated, and NGN-treated groups using a computer-generated random-number list. In contrast, vehicle-injected rats served as the non-diabetic control group.
2.7. Sampling and Biochemical Investigations
After the end of experiments, the overnight fasted rats were euthanized by i.p. injection of thiopental (70 mg/kg). Blood samples were collected through the tail veins, and Fasting Blood Sugar (FBS) was measured by a glucometer. Blood samples were left to clot for 60 min, and centrifuged at 5000 rpm for 10 min, and an ELISA kit was used to estimate serum insulin level (Elabscience Biotechnology Inc., USA).
The eyes of rats were enucleated, and the lenses were dissected out. The lenses were homogenized in cold potassium phosphate buffer (0.05 M, pH 7.4), and then the homogenates were centrifuged to obtain the supernatant. Colorimetric kits were used to measure Malondialdehyde (MDA), Nitric Oxide (NO), and Total Antioxidant Capacity (TAC) (Biodiagnostic, Egypt), and cleaved caspase-3 activity (R&D Systems, USA). In addition, ELISA kits were used to determine Tumor Necrosis Factor-α (TNF-α) and interleukin-6 (IL-6) (R&D Systems, USA), nuclear factor-κB p65 (NF-κB p65) (Novus Biologicals, USA), and Bax and Bcl-2 (LifeSpan Biosciences, USA).
Additionally, in lens homogenates, aldose reductase activity was measured using the spectrophotometric method previously described [14], and a colorimetric kit was used to assess sorbitol levels (Sigma-Aldrich, USA). Moreover, in lens homogenates, glycated proteins were determined using a previously reported method [15], and total and soluble proteins were quantified using a protein assay kit (Biodiagnostic, Egypt).
2.8. Statistics
Data are presented as mean ± S.E.M. Statistical analyses were performed using GraphPad Prism software (version 6.01). Comparisons among the four experimental groups were conducted using a one-way analysis of variance (ANOVA). When the overall ANOVA was significant, Tukey’s multiple-comparison post hoc test was applied for pairwise comparisons between groups. Prior to running parametric analyses, the underlying assumptions were evaluated: data distribution was assessed for approximate normality by visual inspection of histograms and Q–Q plots, and by the Shapiro–Wilk test, and homogeneity of variances across groups was assessed using Brown–Forsythe/Levene-type tests in GraphPad Prism. In instances where the assumptions required for parametric testing were not satisfied, the corresponding non-parametric approach (Kruskal–Wallis test followed by Dunn’s multiple-comparison test) was considered. Data were screened for obvious data-entry or measurement errors, and no values were excluded unless attributable to a documented technical issue. All statistical tests were two-sided, and a p-value < 0.05 was considered statistically significant.
3. RESULTS
A single injection of STZ (45 mg/kg, i.p.) resulted in a significant elevation (P < 0.05) of FBS, and a significant decrease (P < 0.05) of serum insulin, as compared to the corresponding control values (Fig. 1A). Additionally, STZ administration caused significant increments (P < 0.05) of MDA and NO. A significant decrement (P < 0.05) of TAC in rat lenses, in comparison with the control levels (Fig. 1B). On the other hand, treatment with either HSN (100 mg/kg/day, p.o.) or NGN (50 mg/kg/day, p.o.) for 10 weeks, resulted in significant reductions (P < 0.05) of FBS and lenticular MDA and NO, and significant increases (P < 0.05) of serum insulin and lenticular TAC in STZ-diabetic rats (Fig. 1A and B).
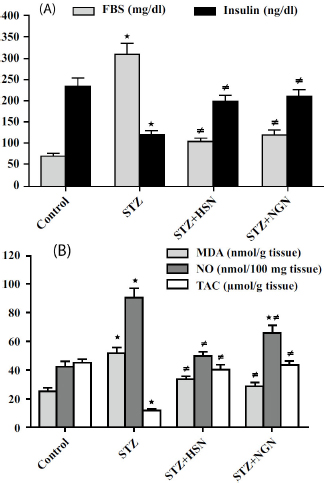
Results of treatment with either Hesperidin (HSN) or Naringenin (NGN) on: (A) Fasting Blood Sugar (FBS) and serum insulin; (B) lenticular Malondialdehyde (MDA), Nitric Oxide (NO), and Total Antioxidant Capacity (TAC) of Streptozotocin (STZ)-diabetic rats. Results are mean ± S.E.M., *P < 0.05 vs. control group, ≠P < 0.05 vs. STZ group.
Moreover, administration of STZ caused significant elevations (P < 0.05) of TNF-α, IL-6, NF-κB p65, cleaved caspase-3, and Bax/Bcl-2 ratio in the rat lenses, as compared to the control values (Fig. 2A-C). Contrarily, the lenticular concentrations of TNF-α, IL-6, NF-κB p65, Bax/Bcl-2 ratio, and cleaved caspase-3 activity showed significant decreases (P < 0.05) in STZ-challenged rats treated with either HSN or NGN (Fig. 2A-C).
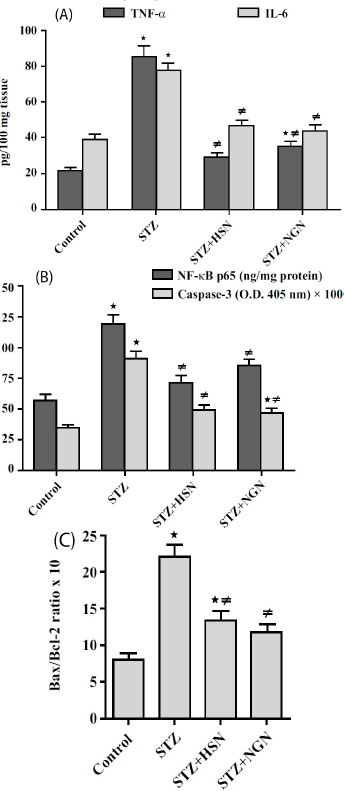
Results of treatment with either Hesperidin (HSN) or Naringenin (NGN) on: (A) Tumor Necrosis Factor-Α (TNF-α) and Interleukin-6 (IL-6); (B) Nuclear Factor-κB p65 (NF-κB p65) and cleaved caspase-3; (C) Bax/Bcl-2 ratio in the lenses of Streptozotocin (STZ)-diabetic rats. Results are mean ± S.E.M., *P < 0.05 vs. control group, ≠P < 0.05 vs. STZ group.
Regarding the parameters of the polyol pathway, significant increments (P < 0.05) of aldose reductase activity and sorbitol concentration were observed in the lenses of rats receiving STZ, in comparison with the control levels (Fig. 3). However, aldose reductase activity and sorbitol concentration were significantly reduced (P < 0.05) in the lenses of STZ-challenged rats, which received HSN or NGN treatment (Fig. 3).
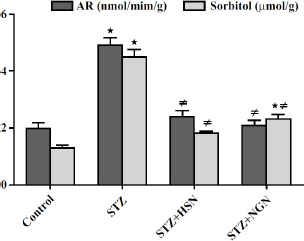
Results of treatment with either Hesperidin (HSN) or Naringenin (NGN) on Aldose Reductase (AR) activity and sorbitol concentration in the lenses of Streptozotocin (STZ)-diabetic rats. Results are mean ± S.E.M., *P < 0.05 vs. control group, ≠P < 0.05 vs. STZ group.
Regarding the results of lens proteins, significant decrements (P < 0.05) of total proteins and soluble proteins, and a significant increment (P < 0.05) of glycated proteins were detected in the lenses of rats receiving STZ injection, as compared to the corresponding control levels (Fig. 4A and B). Again, treatment with either HSN or NGN resulted in significant increases (P < 0.05) in total and soluble proteins, and a significant decrease (p < 0.05) in glycated proteins in lenses of STZ-challenged rats (Fig. 4A and B).
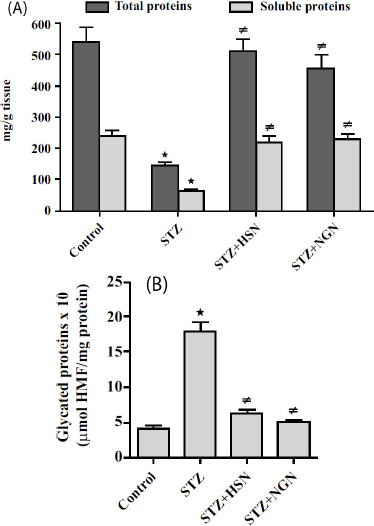
Results of treatment with either Hesperidin (HSN) or Naringenin (NGN) on: (A) total proteins and soluble proteins; (B) glycated proteins in the lenses of Streptozotocin (STZ)-diabetic rats. Results are mean ± S.E.M., *P < 0.05 vs. control group, ≠P < 0.05 vs. STZ group.
Gross lens opacity was visually inspected at the time of dissection, and lenses from STZ-diabetic rats exhibited apparent opacification compared with controls; however, standardized photographic documentation or slit-lamp grading was not performed.
4. DISCUSSION
Consistent with previous studies, the current findings confirm that STZ-induced diabetes in rats leads to pronounced lens damage, characterized by oxidative stress, elevated lipid peroxidation of cellular membranes, and reduced endogenous antioxidant defenses [16]. Nitrosative stress also appears to play a role, with increased production of Reactive Nitrogen Species (RNS), such as Nitric Oxide (NO), contributing to cataract formation in diabetic conditions [17]. Furthermore, oxidative stress and elevated Reactive Oxygen Species (ROS) levels have been shown to activate the NF-κB signaling pathway, particularly by increasing cytoplasmic levels of its active subunit, NF-κB p65. The subsequent translocation of NF-κB p65 into the nucleus triggers the transcription of several pro-inflammatory cytokines, including TNF-α and IL-6 [16, 18]. This inflammatory cascade contributes to the progression of diabetic cataract, exacerbating lens damage. Consistent with these mechanisms, the present study observed notable increases in NF-κB p65, TNF-α, and IL-6 levels in the lenses of STZ-treated rats, indicating heightened inflammation.
HSN and NGN, two flavonoids primarily found in citrus fruits, have been widely recognized for their antioxidant and free radical scavenging properties. One of their key mechanisms involves the suppression of NADPH oxidase, a major contributor to ROS production under oxidative stress conditions [19, 20]. Additionally, these compounds exhibit antinitrosative actions by inhibiting inducible Nitric Oxide Synthase (iNOS), thereby reducing the formation of reactive nitrogen species (RNS) [21, 22]. These findings align with the current study, which showed that treatment with HSN and NGN significantly reduced levels of Malondialdehyde (MDA), a marker of lipid peroxidation, and Nitric Oxide (NO), while increasing Total Antioxidant Capacity (TAC) in the lenses of diabetic rats. It has been well documented that diabetes activates the mitochondrial apoptotic pathway in lens tissue, characterized by increased expression of the pro-apoptotic protein Bax and reduced levels of the anti-apoptotic protein Bcl-2 [23]. This imbalance compromises mitochondrial membrane integrity, allowing cytochrome c to be released into the cytoplasm and triggering caspase-mediated cell death, particularly through caspase-3 activation [24]. Prior studies have demonstrated the antiapoptotic potential of HSN and NGN, which modulate the expression of Bax and Bcl-2, likely through their antioxidative and anti-inflammatory effects [25, 26]. In line with these reports, our study found that both compounds effectively inhibited diabetes-induced activation of the mitochondrial apoptotic pathway, normalizing the Bax/Bcl-2 ratio in lens tissues.
Diabetes has also been shown to upregulate the polyol pathway in the lens, primarily by increasing aldose reductase activity [27]. This enzyme catalyzes the NADPH-dependent conversion of glucose to sorbitol. Additionally, the high glucose concentration in the aqueous humor enters the lens via facilitated diffusion, thereby contributing to intracellular sorbitol accumulation. This hyperosmotic state draws water into the lens, resulting in fiber swelling, liquefaction, and eventual opacification. HSN and NGN have been reported to inhibit aldose reductase activity and reduce sorbitol accumulation in diabetic conditions [28]. Supporting this, the current study demonstrated that treatment with HSN and NGN significantly mitigated aldose reductase activation and limited sorbitol buildup in the lenses of STZ-treated rats. Furthermore, the combined effects of oxidative and osmotic stress promote non-enzymatic glycation of lens proteins, a key contributor to diabetic lens opacity. The interaction of glucose metabolites with amino acid residues leads to aggregation and cross-linking of lens crystallins [29]. These high-molecular-weight, insoluble aggregates are believed to play a central role in lens clouding observed in chronic hyperglycemia [30]. Our findings reveal that HSN and NGN substantially decreased the accumulation of glycated lens proteins while preserving both total and soluble protein content, likely due to their antioxidative properties and inhibition of the polyol pathway.
In addition to their local lens-protective effects, previous studies and the current investigation highlight the antidiabetic potential of HSN and NGN. These compounds were found to lower fasting blood glucose levels and help maintain pancreatic β-cell integrity and insulin secretion [31, 32]. Such systemic effects may contribute indirectly to the prevention of diabetic cataract by improving overall glycemic control in STZ-induced diabetic rats. Beyond citrus flavonoids, several phytochemicals demonstrate lens- and organ-protective effects through antioxidative, anti-inflammatory, and anti-apoptotic mechanisms in models of toxin- or diabetes-related injury. In STZ-diabetic rats, thymoquinone ameliorated diabetes-induced lens changes and delayed cataractogenesis, supporting the mechanistic plausibility of our findings [33]. In non-ocular ototoxicity, hesperidin mitigated gentamicin-induced injury by suppressing oxidative stress, inflammation, and apoptosis, aligning with our observed reductions in MDA/NO, NF-κB activation, and caspase-3 activity [34]. Similarly, thymol provided antioxidant, anti-inflammatory, and anti-apoptotic protection against gentamicin-induced nephrotoxicity in rats, reinforcing the generalizable cytoprotective profile of plant-derived phenolics [35].
Hesperidin and naringenin were selected because of their established antioxidant, anti-inflammatory, anti-apoptotic, and antidiabetic actions in experimental models, while their lens-protective role in diabetic cataract remains less explored. Future work should directly compare HSN and NGN with other flavonoids, such as quercetin or resveratrol, to clarify their relative efficacy in preventing diabetes-induced lens damage.
Lens epithelial cells play a pivotal role in maintaining lens transparency, and accumulating evidence indicates that they are particularly vulnerable to hyperglycemia-induced injury. Under diabetic conditions, these cells are exposed to increased oxidative and nitrosative stress, activation of the polyol pathway, and pro-inflammatory and pro-apoptotic signaling, all of which contribute to disruption of lens homeostasis and subsequent cataractogenesis. Previous in vitro and in vivo studies on lens epithelial cells have demonstrated that hyperglycemia triggers mitochondrial dysfunction, caspase activation, and abnormal protein modification, supporting their central role as an early target in diabetic cataract development. [11] Our findings on lens biochemical alterations are therefore consistent with the concept that interventions that attenuate these pathways may exert protective effects, at least in part, through preserving lens epithelial cell integrity.
5. STUDY LIMITATIONS
This study has several limitations. First, formal lens opacity grading (e.g., slit-lamp scoring or photographic documentation) was not performed; therefore, the conclusions are restricted to lens biochemical protection rather than definitive anti-cataract efficacy. Second, lens epithelial cell–specific or histological analyses were not carried out, and future studies should include a detailed assessment of lens epithelial damage and structural changes. Third, no a priori power calculation was performed, and the relatively small group size may limit the statistical power for some endpoints. An additional limitation is the absence of non-diabetic rats treated with hesperidin or naringenin. Therefore, the potential effects of these flavonoids on normal lens biochemistry or possible toxicity in non-diabetic conditions could not be evaluated in this study. Finally, the pharmacokinetics and bioavailability of hesperidin and naringenin in serum or lens tissue were not evaluated, and potential off-target effects of streptozotocin on the lens cannot be completely excluded.
CONCLUSION
The findings of this study show that hesperidin and naringenin significantly attenuated biochemical lens alterations in streptozotocin-induced diabetic rats. Their protective effects were associated with reduced oxidative/ nitrosative stress, inflammation, apoptosis, polyol pathway activation, and protein glycation, along with improved glycemic status. These results support a lens-biochemical protective effect in this experimental model; however, further studies using standardized cataract grading, histology, and pharmacokinetic assessment are required before clinical relevance can be inferred.
AUTHORS’ CONTRIBUTIONS
It is hereby acknowledged that all authors have accepted responsibility for the manuscript's content and consented to its submission. They have meticulously reviewed all results and unanimously approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| DM | = Diabetes Mellitus |
| STZ | = Streptozotocin |
| HSN | = Hesperidin |
| NGN | = Naringenin |
| FBS | = Fasting Blood Sugar |
| MDA | = Malondialdehyde |
| NO | = Nitric Oxide |
| TAC | = Total Antioxidant Capacity |
| TNF-Α | = Tumor Necrosis Factor-Alpha |
| IL-6 | = Interleukin-6 |
| NF-ΚB P65 | = Nuclear Factor-Kappa B P65 |
| BAX | = BCL-2–Associated X Protein |
| BCL-2 | = B-Cell Lymphoma 2 |
| AR | = Aldose Reductase |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The experimental protocol was approved by the Scientific Research Committee of the Faculty of Medicine, Al-Baha University, and all procedures were conducted in accordance with the institutional and national guidelines for the care and use of laboratory animals. (approval number: REC/SUR/BU-FM/2024/21). Male Sprague-Dawley rats (approximately 200 ± 10 g) were housed under standard laboratory conditions (24 ± 1 ºC, 45% humidity, 12 h light/dark cycle) with free access to chow and tap water, and were allowed to acclimatize for 7 days before the start of the experiment.
HUMAN AND ANIMAL RIGHTS
This study adheres to internationally accepted standards for animal research and follows the 3Rs principle. The ARRIVE guidelines were employed for reporting experiments involving live animals, promoting ethical research practices.
The animal experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
The author would like to thank the technical staff of the animal facility and the laboratory team at the Faculty of Medicine, Al-Baha University, for their assistance during the experimental work and sample processing.


